Turnkey Cleanroom Solutions
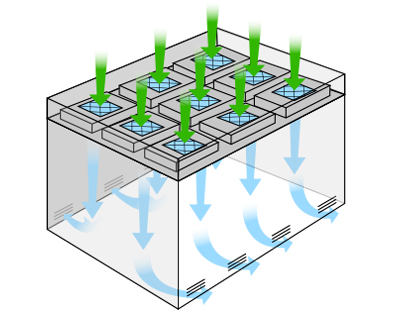
Most Common: Single-Pass Cleanrooms
Single-pass cleanroom systems, also referred to as 'once-through', move air through mounted HEPA filter units within a ceiling grid system. Air is drawn from an adjacent, conditioned space into the cleanroom, and then expelled from near-floor mounted grilles without recirculation. Conveniently, the air expelled from the cleanroom also improves the integrity of immediately surrounding as the cleanroom intakes and filters air outwardly.
Single pass cleanroom systems provide one of the most economical solutions for cleanroom construction. Generally, cleanroom spaces under 400 square feet allow simple recirculation of adjacent ambient air and without succumbing to the heat loads of HEPA fan filter motors, light fixtures, process equipment, air change rates, and number of personnel in the cleanroom. Therefore, single-pass system use is ideal when the control of air temperature and humidity is less critical.
If existing cooling equipment is not sufficient for temperature control, the cost of a single-pass system quickly inflates. Designing the right cleanroom, the first time, is crucial for a long-term return on investment. It is not uncommon for air temperatures within a single-pass air flow cleanroom to reach 10°F higher than ambient air with up to 85% relative humidity. This is problematic for many reasons, including the comfort of users and environmental conditions which promote the growth of fungus and microbes.
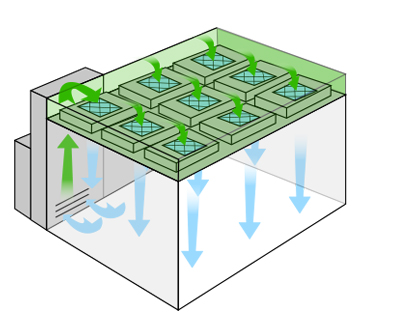
Recirculating Air Flow Cleanrooms
Recirculating cleanroom systems also use fan powered HEPA filtration systems, but instead of drawing air directly from an adjacent environment, a sealed plenum prevents unfiltered bypass air from entering the cleanroom and supplies pre-conditioned air. Recirculating systems provide tighter control of temperature and humidity with more precision and efficiency.
The decision to use a recirculating versus a single pass cleanroom system is more dependent precision of temperature and humidity control than particulate levels. Because temperature and humidity levels have a direct relation, failure to properly tame moisture or ambient air temperature is unacceptable for the fabrication of moisture or ESD sensitive devices such as wafer fabs or semiconductor chip.
Request a Quote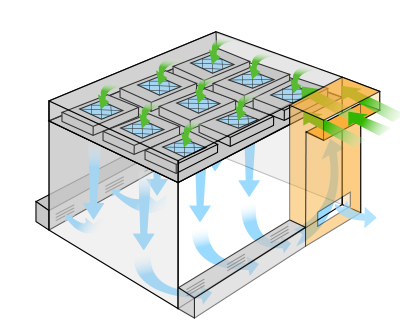
Negative Pressure Cleanrooms
Negative pressure designs prevent contaminants from entering the surrounding area. For processes which include biohazardous, corrosive, flammable, or explosive materials, negative pressure cleanrooms, with the combination of air handers or HEPA filters, exhaust contaminated air to a safe location. Common applications for a negative pressure cleanroom include powder filling, general clean labs, and bio-hazards.
Request a Quote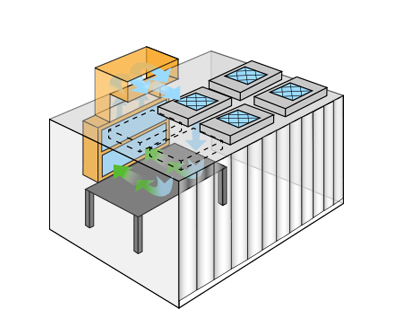
Powder Containment Cleanrooms
Softwall, plastic strip exteriors allow carts or large items to pass easily between spaces. An arrangement of fan filter units, exit filters, and air handlers provide a clean zone adaptable by use or application.
Powder containment systems accommodate neutral, positive, and negative pressure cleanroom configurations. Neutral pressure powder containment designs protect the area outside lab, positive pressure protects products or devices, and negative pressure rooms protect personnel.
Purity testing, weighing, packaging, and chemical analysis applications all benefit from the use of powder containment cleanrooms. HEPA filtration modules draw chemicals away from operators with 99.9% efficiency.
Request a QuoteTurnkey Cleanroom Solutions by Application
Do you need a complete delivered solution? Browse our turnkey cleanrooms for microelectronics or medical device manufacturing, cGMP, USP 797 or 800 compounding, IV compounding, and other applications. Learn about our planning, design, engineering, construction, assembly, and certification services.
Medical Device Manufacturing
The cleanroom requirement for medical devices is based on path to product and process validation, with respect to sterilization and biocompatibility requirements. Learn more.
Microelectronics Manufacturing
Manufacturers of semiconductors, displays, HDDs, wafer chips and more have long recognized the relationship between cleanroom air quality and final yields. Learn more.
GMP (Good Manufacturing Practices)
cGMP sets forth the requirements applicable to the design of facilities used in the manufacture, processing, packing, or holding of a drug product. Learn more.
USP 797 Compounding
Ideal for positive pressure compounding of non-hazardous materials including TPN, injections, eye-drops, infusion, syringes, salves, oils, and more. Learn more.
USP 800 Hazardous Drugs
USP 800 grade environments mitigate the risk for harm to both the environment and workers when compounding hazardous chemicals. Learn more.
Agriculture & Grow Rooms
Modular cannabis cultivation rooms have many advantages over traditional grow rooms. Mother, clone, and flower rooms flourish when room-to-room temperature and humidity are regulated. Learn more.



