Differences Between USP 795, 797, and 800 Cleanrooms
For some healthcare facilities, upgrading or retrofitting a cleanroom for USP 800 compliance requires infrastructural and mechanical modifications. Small modifications may include equipment upgrades while more extensive upgrades may include expanding cleanrooms, new ductwork and dedicated exhaust systems.
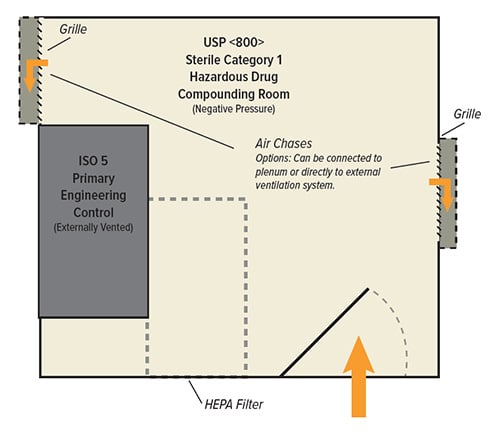
Negative pressure environments mitigate the risk for harm to both the environment and workers when compounding or packaging antineoplastic drugs, hormonal therapies radiopharmaceuticals, or during the research of pathogens, viruses, and other harmful bacteria.
Positive pressure ensures that in the case of a breached barrier, the space maintains sterility. An direct compounding area with an ISO Class 5 laminar flow device requires no less than 30 ACH (air changes per hour). Positive pressure rooms allow compounding of TPN (Total Parenteral Nutrition), antibiotic injections, eye-drops, infusion, syringes, salves, oils, and more.
Difference Between USP 797 and USP 800 Cleanrooms
The purpose of USP 797 is a general protection of sterile compounds and spaces from contamination. USP 800 expands controls for the protection of workers and environments against hazardous drug compounds. In contrast to USP 797, which only remedies sterile compounding activities, USP 800 takes a 360-degree approach for processing hazardous drugs.
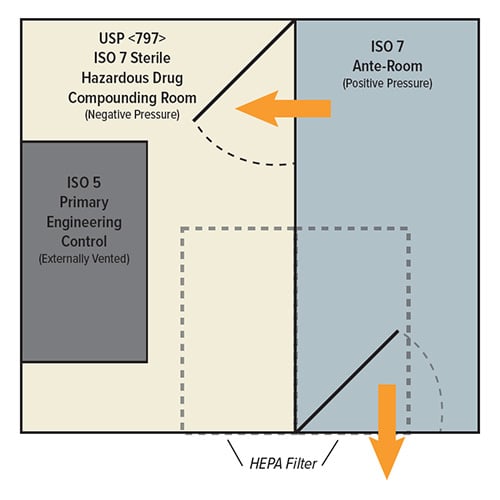
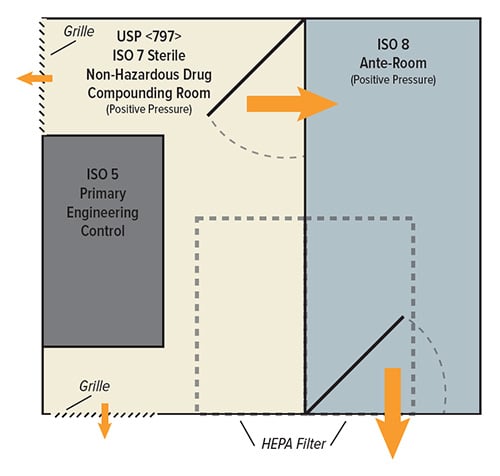
Changes from USP 795 to USP 797
USP 797 builds upon the same 795 requirements for NHDs, with the additional requirement of an externally vented laminar flow hood as a primary engineering control (PEC) for HDs. USP 797 also expands environmental controls with positively pressured ISO 5 PECs, ISO 7 buffer rooms, and ISO 8 ante-rooms. USP 797 also enforces a separation of spaces for the compounding of HDs and NHDs including water column specifications for pressure differentials.
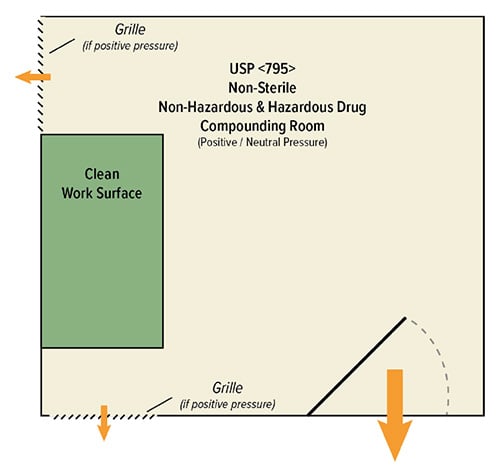
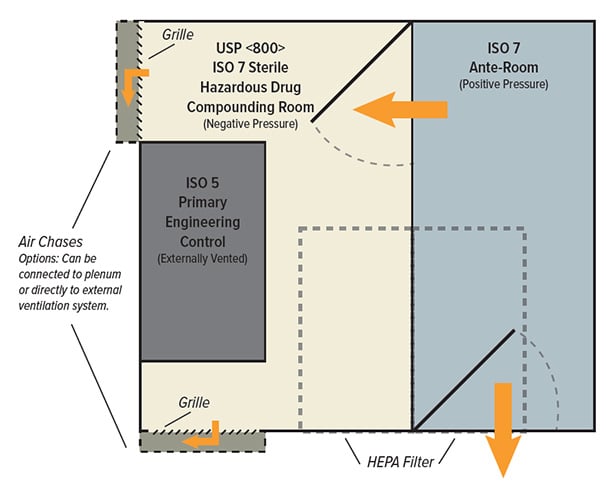
Significant Challenges for Retrofitting USP 795 Cleanrooms
Hazardous drugs (HDs) or non-hazardous drugs (NHDs) do not differ in facility requirements under USP 795. Non-sterile compounding areas simply require separation from sterile compounding areas and climate control. Non-sterile HDs and sterile HDs within shared spaces previously met USP 795 requirements before USP 797 revisions.
USP 800 Considerations
USP 800 engineering controls and ante-rooms may present challenges for stressed air-handling systems. Additional storage and auxiliary space for separate storage of hazardous and nonhazardous drugs also requires consideration. USP 800 does not allow storage, unpackaging, or manipulation of HDs in positive pressure areas, nor does it allow the USP 797 exemption for low volumes of HDs to be compounded in a non-negative pressure room. While USP 800 does not address pressure monitoring for nonsterile preparations, a differential pressure gauge is key when monitoring a negative pressure environment.
 USP 800 Requirements for Unpacking Hazardous Drugs
USP 800 Requirements for Unpacking Hazardous Drugs
Facilities unpacking hazardous drugs within a negative pressure room do not require procedural changes to meet USP unpacking area requirements. Facilities unpacking HDs within positive pressure ante rooms or facility spaces will require reconsideration.
USP 800 dictates a clear separation for the storage of non-hazardous and hazardous drugs. An exception is made for HDs contained within their final unit dose or unit-of-dose packaging and appropriately labeled. Drug inventory rooms now require negative pressure storage with external ventilation and a minimum of 12 ACH. Learn more.

